Buprenorphine Facts
What is Buprenorphine?
- Buprenorphine is a partial opioid agonist that binds to opioid receptors in the brain, reducing cravings and withdrawal symptoms when opioids are stopped.
- Buprenorphine does not have the same effects as full agonist opioids, especially when combined with naloxone.
- Buprenorphine is used to treat both opioid use disorder (OUD) and chronic pain. In this continuing education program, we focus on the use of buprenorphine to treat OUD.
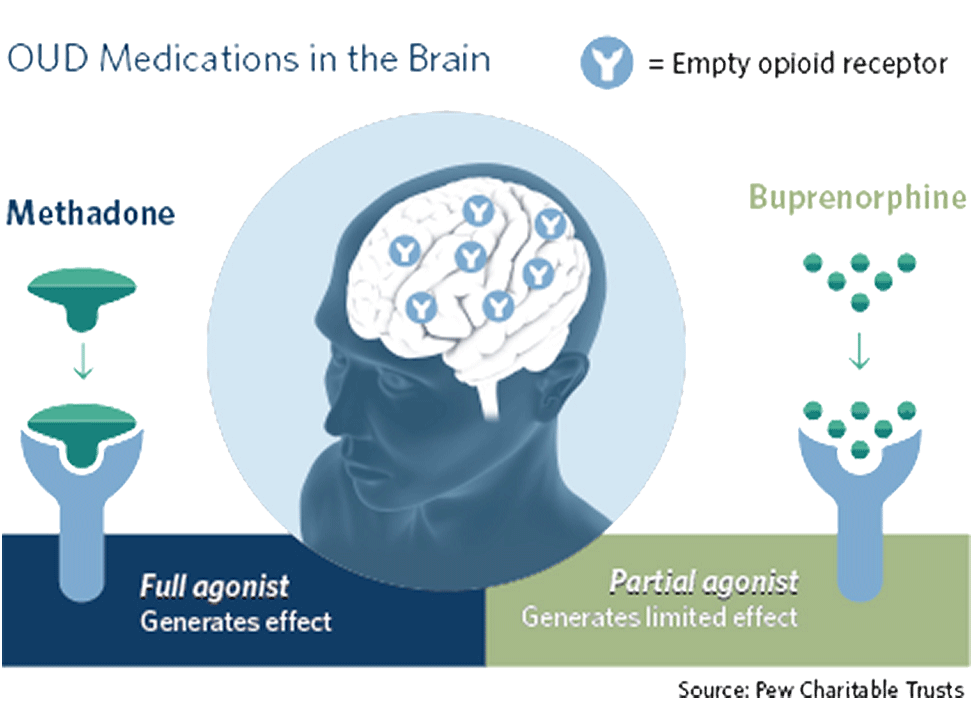
How is Buprenorphine Prescribed?
General Guidelines
| Statement | True or False | Result | |
|---|---|---|---|
| Buprenorphine is usually prescribed as a short-term medication. | |||
| Buprenorphine has been shown to be diverted more frequently than oxycodone and hydrocodone. | |||
| Providers are required to be registered with the DEA to prescribe buprenorphine. | |||
| Providers are required to have a DEA X-waiver in order to prescribe buprenorphine. | |||
| The DEA places limits on the number of patients that advanced practice providers are able to see and prescribe buprenorphine. | |||
| Telehealth providers can prescribe buprenorphine. | |||
Reasons for Diversion
Inability to obtain prescribed buprenorphine can contribute to diversion.2, 3, 7

Monoproduct Buprenorphine
It is important for the pharmacists to communicate with the prescriber to document the rationale behind the monoproduct prescription on the pharmacy record.
It may be appropriate for a patient to receive a monoproduct buprenorphine instead of combination product buprenorphine/naloxone for reasons such as:
Naloxone intolerance such as nausea, vomiting, or sensitivity to buccal films.8, 9
Monoproduct buprenorphine may be less costly when compared to the combination product with naloxone.
Use in pregnant populations – although the combination product buprenorphine/naloxone may also be used. 10, 11, 12
Injectable monoproduct is less likely to cause tooth decay than combination tablets and buccal films. 13
Insurance may only cover a certain formulation.
Patient assistance may only be available for a certain formulation.
For all referenced materials and a comprehensive list of buprenorphine product formulations available as of January 2025, please see Resources.

